
Gold is malleable, portable, nontoxic, beautiful and rare. Those qualities have made it an excellent currency over the centuries.
But there’s another metal with all those properties and more—and it’s much more valuable.
It’s called rhodium, part of the platinum group of metals. The name comes from the Greek rhodon, meaning rose, for the rosy color of rhodium compounds.
It’s abundant in other parts of the universe but vanishingly rare in Earth’s crust—just one part in 200 million. We mine 100 times more gold each year.
Eighty percent of the world’s rhodium comes from South Africa. And 80 percent of annual supply is consumed by the auto industry.
That’s because, like all metals in the platinum group, rhodium is an excellent catalyst: it controls and encourages chemical reactions.
In your car, that means it’s used in the catalytic converter.
Exhaust flows through a metal honeycomb lightly spattered with rhodium and other platinum group metals. They turn dangerous nitrogen oxides and carbon monoxide into nontoxic nitrogen gas, carbon dioxide and water vapor.
Rhodium’s catalytic properties are in demand in other industries too.
There’s even an experimental technology that uses solar energy to turn carbon dioxide and water into high energy fuels using rhodium as a catalyst.
Background
Synopsis: The most valuable metal you own may not be in your jewelry box; it may be in your car! Rhodium is one of the key catalysts that change noxious gases to cleaner emissions in automotive catalytic converters. Recently, more stringent environmental measures have caused increased demand, while supply chains have struggled in the wake of the COVID 19 pandemic. As a result, by March 2021 the value of rhodium soared to more than 16 times that of gold by weight.
- When asked about Earth’s most valuable metals, most of us think of gold (Au), but rhodium (Rh) is much more valuable.
- Gold has been used as currency through the ages because it is moderately rare, nontoxic, nonreactive even in acids, malleable, portable, and beautifully distinctive in appearance.
- It has societal value because cultures historically depended upon it for trade.
- Rhodium has many of the same properties, with an added catalytic superpower.
- In March 2021, the value of rhodium increased dramatically to $28,775 per troy ounce ($925 per gram), which was 16.5 times the value of gold (traded at $1,739 per troy ounce ($56 per gram) on that day).
- By the end of 2021, the value of rhodium had dropped to eight times the value of gold as automakers decreased demand when they slowed or shut down production because of a pandemic-related semiconductor shortage, but it was still Earth’s most valuable metal.
- Rhodium has an atomic number of 45. Like gold, it is considered a noble metal because it is nontoxic and nonreactive, resisting corrosion and oxidation even at high temperatures.
- Within the noble metals, rhodium (Rh) is a member of the platinum group along with ruthenium (Ru), palladium (Pd), osmium (Os), iridium (Ir), and platinum (Pt).
- These six elements are superior catalysts; they expedite and control the rates of many important chemical reactions.
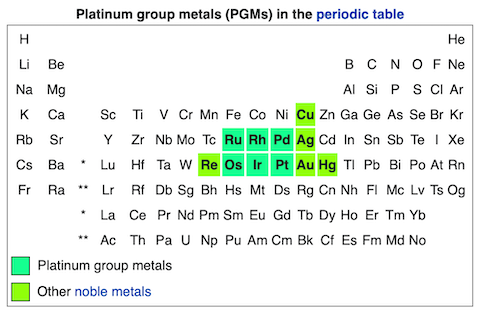
- Platinum group elements (PGEs) are exceedingly rare in Earth’s upper crust, but they have higher concentrations in Earth’s mantle and are abundant in the universe.
- PGEs are normally found mixed together in ore, so rhodium is typically a byproduct of mining for gold, platinum, palladium and silver, and small quantities are also produced during nickel and copper smelting.
- Deposits occur in dark-colored mafic- and ultramafic-layered igneous intrusions and mineralized sills, as well as in primitive planetary material found near metallic impact craters.
- More than 90% of these elements are found in South Africa’s Bushveld Complex, Zimbabwe’s Great Dyke, and Russia’s Norilsk region.
- The 1.85-billion-year-old Sudbury Impact Structure is another source of PGEs.
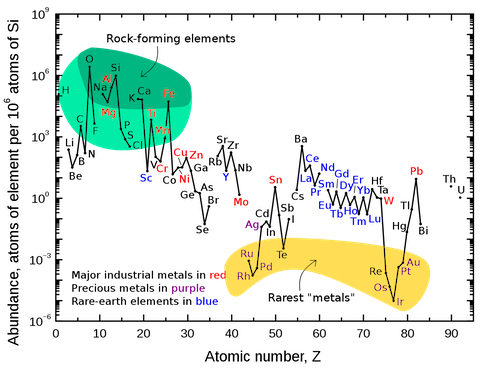
- Rhodium is the rarest metal in this group, representing only 1 part per 200,000,000 in Earth’s crust. Miners produce about 100 times more gold each year than rhodium.
- Its name comes from the Greek word for rose rhodon because of the rosy color of some of its compounds.
- More than 80% of demand for rhodium comes from the automotive sector.
- South Africa currently produces more than 80% of the global annual production of rhodium.
- The United States imports 90% of its PGEs, and they are in high demand for modern technologies.
- They are required in the manufacturing processes for glass, fertilizer, and products like cancer drugs, and can be found in jewelry, electronics, and fuel cells in addition to automotive catalytic converters.
- New technologies may depend upon these catalysts. A promising new process that uses the sun’s energy to turn carbon dioxide and water into high-energy fuels using cerium oxide requires rhodium as a catalyst (but that’s a story for another EarthDate episode).
- Catalytic converters treat automotive exhaust emissions by turning toxic nitrogen oxides and carbon monoxide into less toxic N2 gas, carbon dioxide and water.
- In a catalytic converter, exhaust gases are forced through a honeycomb spattered with three of the most catalytic PGEs (rhodium, palladium and platinum).
- These critical catalysts accelerate key reactions that enable simultaneous oxidation and reduction to take place, rendering noxious gases less harmful.
- Rhodium is the most active of the three.
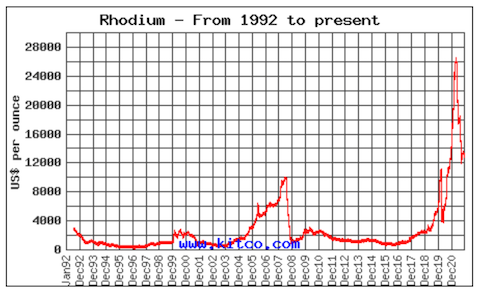
- Rhodium’s incredible value was all about supply and demand in 2021.
- As governments tightened automotive emissions rules in 2018–2019, the demand for catalytic converters increased dramatically. Meanwhile, amazing new technologies are coming online that require rhodium’s catalytic power.
- At the same time, supply has been challenged because not enough rhodium is being produced as a byproduct of PGE mining.
- In addition, Covid restrictions have limited the mining workforce in South Africa.
- However, once automotive demand decreased as car production slowed or stopped because of pandemic-related semiconductor shortages, the value of rhodium dropped dramatically.
- In the future, electric cars will run on batteries requiring different toxic metals, but they won’t need catalytic converters. Hydrogen fuel cells require PGEs to function so demand will continue.
- Unfortunately, the value of rhodium has led to illegal trade in the metal, targeting catalytic converters, especially in hybrid vehicles.
- Hybrid cars deplete catalysts more slowly, and earlier hybrid models had larger quantities of PGEs speckled on the honeycomb structures of their converters, so they are the target of most catalytic converter thefts.
- Thieves usually jack the cars up, saw off the converter, and jack the cars down in a matter of minutes. Without the catalytic converter, cars exhausts produce a shockingly loud roar.
- Replacing a catalytic converter may cost more than $1000. Keeping cars in locked garages or installing metal plates to prevent theft are preventative options.

