
Lithium, the lightest metal, has transformed our rechargeable-battery technology. But it can also be transformative in the human mind.
Its powers were recognized by early cultures, from the Greeks to Native Americans. And for decades it has been used as a pharmaceutical drug.
Lithium is highly reactive—it’s one of the few metals that will easily burn. If you try to put it out with water, it reacts violently, as it splits off hydrogen gas, which also burns.
When lithium burns, it creates a crimson light—which you’ve seen whenever you’ve watched a fireworks show. Lithium makes their signature red color.
Lithium is so reactive it’s typically stored in mineral oil. But when it enters the human body, often as a much less reactive lithium salt, its incendiary personality does a 180.
In a process we don’t completely understand, it seems to moderate the flow of electricity and promote the overall health of our nervous system. The result is a calming effect.
This has led doctors to prescribe lithium as a successful treatment for bipolar disorder. Studies have even found that cities and towns with higher levels of lithium salts in their groundwater have markedly lower levels of depression, suicide, and violent crime.
A very surprising fact—which has led some scientists to recommend that lithium be considered an essential nutrient. It may be time to take a closer look at this ancient remedy.
Background
Synopsis: Lithium is an extremely reactive element that also has an amazing calming effect on the human brain. It is regularly prescribed for bipolar disorder, possibly buffering the flow of ions or enhancing the growth of neurons in the brain. Higher concentrations of lithium in groundwater have been correlated with decreased incidence of suicide, homicide, and assault.
- Lithium is a very special element, with its very small size and extreme reactivity.
- Lithium is a soft, silver-white metal with the atomic number 3; only hydrogen and helium have less mass, and they are gases.
- Lithium was one of the first elements to be synthesized—13.7 billion years ago in the Big Bang along with hydrogen, helium, and beryllium. But because of its relative nuclear instability, lithium is less common in the solar system than many other chemical elements.
- Lithium ore is produced from prehistoric brine deposits that evaporated repeatedly to concentrate lithium eroded from nearby mountains and hydrothermal deposits into huge salt flats over more than 30,000 years of geologic time.
- The largest of these salt flats are found in the Atacama Desert of Bolivia and Chile.
- Lithium is also produced from minerals found in igneous pegmatites.
- Lithium belongs to the alkali metal group of chemical elements, which are never found in their pure form in nature.
- These elements are very reactive because they all have a single energetic electron orbiting in their outermost shell that is eager to react with other atoms.
- Lithium metal is so reactive, it is typically stored in mineral oil.
- When combined with water, it reacts violently as it evolves hydrogen gas.
- Lithium is flammable. Lithium powder burns crimson red and is the key ingredient in red fireworks.
- Even lithium’s nucleus verges on instability, with very low binding energies. In the 1950’s, one of its isotopes was used in thermonuclear weapon research.
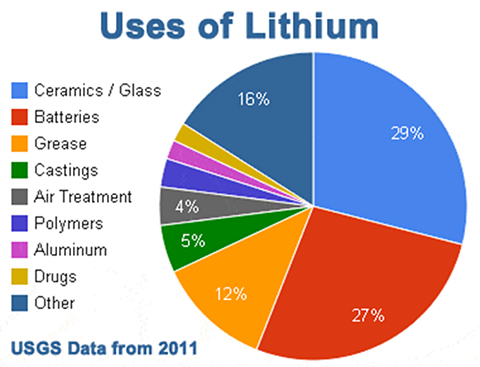
Credit: USGS - Despite its reactivity, the curative powers of lithium were well known by Greek, Roman, and Native American peoples to calm gout inflammation as well as mania.
- By the late 19th century, Lithia Springs, Georgia, was a famous health destination visited by Mark Twain and Presidents Grover Cleveland, William Howard Taft, William McKinley, and Theodore Roosevelt.
- The soft drink 7 Up contained lithium citrate until 1948.
- Lithium is regularly prescribed for bipolar disorder, although how it works is a bit of a mystery. One theory is that our nervous systems rely on flows of positively charged ions of sodium and potassium, and that lithium ions may buffer swings in these flows believed to cause bipolar disorder. Another suggests that lithium might protect or enhance the growth of neurons. Overdosing can be toxic or lethal.
- Lithium is found in groundwater in concentrations thousands of times smaller than medical dosages. Since the 1970’s, research from around the globe shows that higher concentration of lithium in groundwater correlates with decreased incidence of suicide, homicide, and assault and coincides with “beneficial clinical, behavioral, legal and medical outcomes.”

