
Earth is mostly covered with water, and most of that is ocean. Only 2.5 percent is fresh water.
Of that, 99 percent is locked up in glaciers and underground aquifers. That leaves just 1 percent of Earth’s fresh water on the surface.
From all that salt water, how does this tiny fraction of surface fresh water come to be? It’s a process of natural distillation.
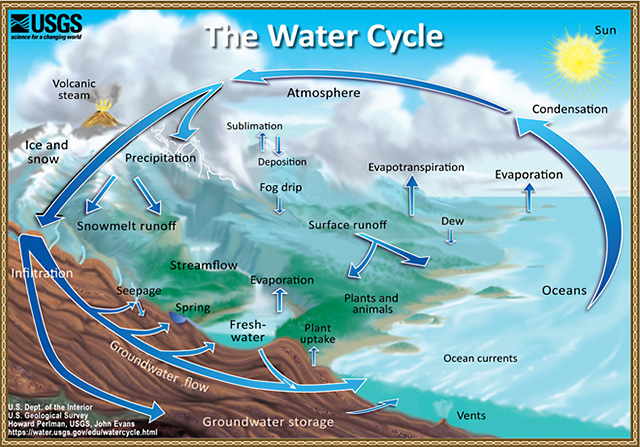
Heat and wind turn seawater into water vapor. In the phase change from liquid to gas, water leaves salt and all impurities behind.
In the atmosphere, water condenses on airborne particles and rains down again.
Since Earth is mostly ocean, most rain falls in the ocean. The part that falls on land flows downhill, eventually into rivers that carry it back into the sea, to become salty again.
That brief, shining moment as surface fresh water has made virtually all land-based life possible, for hundreds of millions of years.
Here’s a practical tip: If you ever find yourself in a dire situation with no fresh water, remember this distillation process.
First, never drink seawater; it’s four times saltier than blood. To neutralize it, your organs will draw water from the rest of your body, leading to rapid dehydration.
Instead, find a way to make your own cloud. Trap rising water vapor, allow it to condense on a surface, and drain it into something that you can drink from.
Background
Synopsis: Of all the water that covers 71 percent of Earth’s surface, 97.5 percent is salt water and only 2.5 percent is fresh. How does Earth maintain its supply of fresh water? If you needed drinking water in an emergency, how could you turn salt water into fresh water?
- Earth’s fresh water exists because the water cycle removes salt, or desalinates, seawater constantly.
- The sun drives winds and waves that churn the seas and cause evaporation of H20 molecules, leaving salts, organics, and other chemicals behind.
- The pure water vapor rises into the atmosphere and condenses into clouds as it encounters cooler conditions. It then falls back to Earth as fresh water, filling rivers and streams if it falls over land.
- The water cycle is essentially a distillation process—evaporating, condensing, and collecting water molecules.
- A whopping 99 percent of Earth’s fresh water is either locked up in glacial ice (around 66 percent) or hidden away in underground aquifers (around 33 percent). Earth’s surface water, in rivers and lakes, represents only 1 percent of Earth’s fresh water—which is just 0.025 percent of all of Earth’s water.
- All land-based life on Earth evolved and lived off of this tiny fraction of Earth’s water for hundreds of millions of years. Through this evolution, humans became dependent on fresh water for life.
- Sources of fresh water dictated where humans could live and was often the driving force behind Paleolithic migrations.
- Man first learned to augment intermittent surface freshwater supplies by digging wells to access fresh groundwater resources. The oldest water wells we know of are early Neolithic wells located in Cyprus that are 10,500 years old. Today, about 20 percent of water used in the United States comes from groundwater.

- Seawater is 3.5 percent salt, but human blood is usually about 0.9 percent salt.
- Our kidneys filter and clean our blood, producing urine to remove excess salt from the bloodstream. However, human kidneys can only create urine from liquids with less than 2 percent salt.
- To dilute ingested salty seawater, the kidneys grab fresher water from other systems in the body, so the body ends up expelling more fresh water to balance the salt, causing thirst, dehydration, and eventually death.
- If you’re ever in a water predicament, remember that distillation is a recipe for personal survival. It is effective for both salty and polluted water.
- Boiling water is important to kill off biological contaminants, but it won’t reduce toxic chemicals in polluted water.
- Collecting and condensing steam as water evaporates separates pure water vapor from any toxins, which become concentrated in the raw water.
- You can boil water in a pot with an inverted lid, and let the condensation drip into a cup hanging suspended from the lid to capture pure water. To avoid contamination, it is important that an air gap exists to separate raw water from the distilled water.

