Credit: Conceptual Image Lab, NASA/Goddard Space Flight Center
The Sahara Desert and the Amazon rainforest are about as different as two places on Earth could be. But they’re tightly linked together in a way you could hardly imagine.
It all started 7,000 years ago, when the Sahara wasn’t a desert at all, but a land of huge lakes. The largest was the mega-lake Chad, bigger than all of our Great Lakes combined.
The mega-lake was home to a huge number of diatoms, algae-like plankton that form the base of the food chain. After algal blooms, billions of dead diatoms would sink to the lake floor.
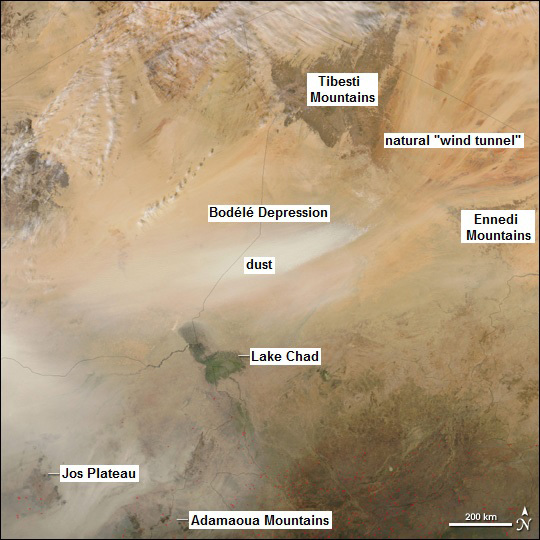
When natural climate change caused the lake to shrink dramatically, it left behind the massive Bodélé Depression, which is now called the “dustiest place on Earth.”
This dust is rich with phosphorous from centuries of dead diatoms and iron from the lakebed. Each winter, winds blow down from the mountains of Saharan Africa and carry it high into the sky.
The dust then blows across the Atlantic in huge plumes and over the Amazon.
There, rising water vapor from the rainforest condenses on the dust particles and rains back down, rich in minerals, like fertilizer from the sky. The phosphorous and iron are essential to the vigorous plant health across the region.
Amazingly, at about the same rate that the Saharan winds carry it in, floods wash previous years’ phosphorous out of the soil, down river, and into the sea.
There, it feeds new algal blooms—completing a circle thousands of miles wide and thousands of years in the making.
Background
Synopsis: What does the dustiest place in the largest desert in the world have to do with the largest rainforest in the world? Howling winds blow phosphorous-rich dust, made up of the remains of tiny microorganisms, 3,000 miles across the Atlantic to fertilize the Amazon rainforest.
- The Sahara is the largest desert in the world, occupying an area about the size of the United States (including Alaska and Hawaii) or China.
- In 5000 BC, the Sahara had a much different climate, and mega-lake Chad was the largest of several Saharan freshwater lakes.
- Lake Chad had a surface area measuring more than 360,000 km2 (140,000 mi2), about the size of the Caspian Sea and larger than the five North American Great Lakes combined.
- The waters of the lake teemed with life, including microscopic organisms called diatoms, a type of algae or phytoplankton that form the base of the food chain.
- After algal blooms, huge volumes of diatoms died and dropped through the water column to the lakebed.
- The mega-lake dried up rapidly, leaving behind the deep Bodélé Depression and today’s Lake Chad, now only 1,350 km2 (520 mi2).
- The Bodélé Depression is called the dustiest place in the world.
- It is in a perfect orientation for strong sustained winds to funnel down into it through nearby mountain ranges on a daily basis at least 100 days per year.
- The howling winds scour away its thick deposits of diatoms that break down into tiny mineral-rich particles of dust enriched in phosphorous. The dust is also enriched in iron from old lakebed.
- Plant growth, respiration, energy transfer, and storage depend on phosphorous, so it is a major constituent of commercial fertilizer. Plants use iron to make an enzyme critical for photosynthesis.
- Satellite imagery shows huge tan-colored plumes of dust that blow from the Sahara almost daily toward the Amazon during Northern Hemisphere winter and shift toward the Caribbean in the summer.
- When abundant water vapor in the Amazon nucleates on the dust particles, mineral-laden rain falls. Water-soluble phosphorous is dissolved in the rain into a form that is easily taken up by plants.
- Meanwhile, water-soluble phosphorous and other minerals are used by plants then washed out of the soil and transported by the Amazon River system back to the sea, where they fertilize ocean algal blooms.
- The amount of phosphorous brought to the Amazon by dust roughly equals the amount that is lost to the sea each year. Mother Nature perfectly balances the Amazon’s need with the Sahara’s gift.
- Lightning from Amazon thunderstorms brings water-soluble nitrogen to plants.
- Gaseous nitrogen is N2—plants can’t use it in that form.
- Lightning breaks the N2 molecule apart, creating the high-temperature and high-pressure environment needed to enable nitrogen to react with oxygen to form nitrogen oxide (NO).
- NO dissolves in rainwater so that plants can use the nitrogen, another key component of commercial fertilizers.